Global mRNA Targeting Biopolymers Market Witnesses Robust Growth as Europe Emerges Strong with BioNTech, CureVac & Roche
Rising demand for GMP-certified delivery systems, oncology pipeline growth, and regulatory innovation position Germany as Europe’s mRNA biopolymer leader
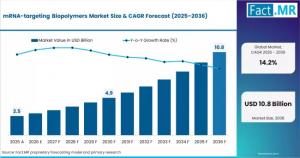
ROCKVILLE, MD, UNITED STATES, April 17, 2026 /EINPresswire.com/ -- According to the latest analysis by Fact.MR, the global mRNA-targeting biopolymers market is valued at USD 2.2 billion in 2025 and is projected to reach USD 2.5 billion in 2026, expanding to USD 9.5 billion by 2036. The market is expected to grow at a CAGR of 14.2%, generating an incremental opportunity of USD 7.0 billion over the forecast period.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14739
The market is undergoing a structural transformation driven by the shift from prophylactic vaccines to therapeutic mRNA applications, particularly in oncology and rare diseases. Germany is emerging as a critical European hub due to its advanced biopharmaceutical infrastructure, strong regulatory alignment with EMA standards, and deep integration into global clinical supply chains.
Quick Stats
Market Size (2025): USD 2.2 Billion
Market Size (2026): USD 2.5 Billion
Forecast Value (2036): USD 9.5 Billion
CAGR (2026–2036): 14.2%
Incremental Opportunity: USD 7.0 Billion
Leading Segment: Lipid-Polymer Hybrids (42% share)
Leading Application: Prophylactic Vaccines (46% share)
Leading Region: Asia Pacific (fastest growth)
Germany CAGR: 12.9%
Key Players: Evonik Industries AG, Merck KGaA, Lonza Group, Thermo Fisher Scientific, Maravai LifeSciences
Executive Insight for Decision Makers
The market is shifting from volume-driven vaccine supply to high-value, precision-targeted therapeutic delivery systems.
Strategic Shift:
Demand is consolidating around GMP-certified, clinically validated biopolymers with organ-targeting capabilities.
What Stakeholders Must Do:
Invest in lipid-polymer hybrid platforms with validated targeting performance
Align with EMA regulatory frameworks for ATMP compliance
Build end-to-end capabilities from synthesis to clinical-grade delivery
Risk of Inaction:
Suppliers lacking GMP certification and documentation are being excluded from late-phase clinical procurement, resulting in lost high-margin opportunities.
Market Dynamics
Key Growth Drivers
Expansion of mRNA oncology and rare disease pipelines
Rising demand for GMP-grade delivery systems
Government-backed biomanufacturing investments globally
Increasing adoption of organ-targeted delivery technologies
Key Restraints
Complex GMP documentation and validation requirements
High concentration of platform intellectual property ownership
Limited scalability of natural biopolymer supply
Emerging Trends
Shift toward non-liver targeted delivery systems
Integration of delivery materials with CDMO services
Growth of personalized mRNA therapeutics
Premiumization of clinical-grade biopolymers
Segment Analysis
Leading Segment:
Lipid-polymer hybrids account for 42% market share (2026) due to superior stability and delivery efficiency.
Fastest-Growing Segment:
Therapeutic mRNA applications, especially in oncology, are witnessing exponential growth.
By Application:
Prophylactic Vaccines – 46%
Therapeutics (Oncology & Rare Diseases) – fastest growth
By End User:
Biopharma & Biotech – 47% share
Strategic Importance:
Advanced delivery systems are becoming the core differentiator, replacing traditional API-centric competition.
Supply Chain Analysis (Critical Insight)
Structure Overview:
Raw Material Suppliers:
Specialty chemical providers supplying lipids, polymers, and excipients
Manufacturers / Producers:
Companies producing GMP-certified biopolymers and hybrid systems
Distributors / CDMOs:
Integrated players offering formulation + manufacturing + delivery solutions
End Users:
Biopharmaceutical companies
Vaccine developers
Research institutions
Who Supplies Whom:
Raw material suppliers → Biopolymer manufacturers
Biopolymer manufacturers → CDMOs / Biopharma companies
CDMOs → Clinical-stage and commercial drug developers
Germany Insight:
Germany-based players are deeply embedded in clinical supply chains, supplying validated materials directly to late-stage trials, ensuring long-term contracts and high switching barriers.
Pricing Trends
Commodity vs Premium:
Commodity biopolymers face pricing pressure
GMP-certified, clinically validated materials command premium pricing
Key Price Influencers:
Regulatory compliance (EMA standards)
Clinical validation data
Raw material costs
Demand from oncology pipelines
Margin Insights:
High margins are concentrated in clinical-grade delivery systems, while research-grade materials remain price-sensitive.
Regional Analysis
Top 5 Countries by CAGR (2026–2036)
China – 15.1%
India – 14.8%
United States – 14.2%
South Korea – 13.5%
Germany – 12.9%
Germany Market Insights
Strong biopharma R&D ecosystem
Leadership in EMA-compliant manufacturing
Increasing demand from clinical-stage mRNA programs
Developed vs Emerging Markets
Developed Markets (Germany, U.S.):
Innovation-led, high-margin, regulation-driven
Emerging Markets (India, China):
Volume-driven, cost-efficient manufacturing expansion
Competitive Landscape
Market Structure: Moderately consolidated at the high-value tier
Key Players
Evonik Industries AG
Merck KGaA
Lonza Group
Thermo Fisher Scientific
Maravai LifeSciences
Arcturus Therapeutics
Genscript Biotech
Precision BioSciences
Sanofi (Translate Bio)
Aximmune Inc.
Competitive Strategies
Platform innovation (targeted delivery systems)
Long-term clinical supply agreements
Integration with CDMO services
Regulatory compliance as a competitive moat
Strategic Takeaways
For Manufacturers
Prioritize GMP-certified production and clinical validation
Invest in hybrid delivery platforms
For Investors
Focus on companies with platform ownership and regulatory alignment
Target firms embedded in clinical-stage supply chains
For Marketers / Distributors
Emphasize performance data and compliance over pricing
Build partnerships with biopharma innovators
Future Outlook
The market is set to transition into a high-value, innovation-driven ecosystem dominated by therapeutic applications.
Expansion of personalized mRNA medicines
Integration of AI-driven formulation design
Sustainability focus in biopolymer sourcing and synthesis
Germany is expected to remain a strategic innovation hub, balancing regulatory rigor with advanced manufacturing capabilities.
Conclusion
The mRNA-targeting biopolymers market is entering a decisive growth phase defined by clinical validation, regulatory compliance, and targeted delivery innovation. Germany’s strong infrastructure and innovation ecosystem position it as a key European leader in this transformation.
For decision-makers, the opportunity lies not in scale alone, but in precision, compliance, and platform differentiation the defining factors of long-term competitive advantage.
Why This Market Matters
mRNA-targeting biopolymers are foundational to the next generation of precision medicine, enabling breakthroughs in oncology, rare diseases, and personalized therapeutics. As the industry evolves, control over delivery systems will determine who leads and who follows in the global biopharmaceutical value chain.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/14739
To View Our Related Report:
IV Bag and Tubing Recycling Market: : https://www.factmr.com/report/iv-bag-and-tubing-recycling-market
In-vitro Human Models Market: https://www.factmr.com/report/in-vitro-human-models-market
Substrate Reduction Therapies Market: https://www.factmr.com/report/substrate-reduction-therapies-market
Bioactive Sphingolipid Market: https://www.factmr.com/report/bioactive-sphingolipid-market
S. N. Jha
Fact.MR
+1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.