Pharmaceutical Microbiology QC Testing Market Growth Projections US$10.84 billion by 2033, Growth in Quality Assurance
Pharmaceutical Microbiology QC Testing Market with Modern Microbiology Testing Techniques (2026)
United States Pharmaceutical Microbiology QC Testing Market (2026): Streamlined Solutions for Sterility and Safety”
AUSTIN, TX, UNITED STATES, March 4, 2026 /EINPresswire.com/ -- Market Size Growth (2026)— DataM Intelligence 4Market Research LLP
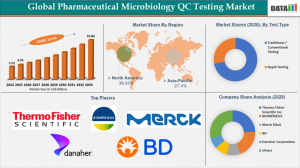
pharmaceutical microbiology QC testing market was valued at US$3.52 billion in 2024, is projected to increase to US$4.05 billion in 2025, and is anticipated to reach US$10.84 billion by 2033, expanding at a CAGR of 13.1% between 2026 and 2033.
Request Executive Sample Report (Get Higher Priority for Corporate Email ID):– https://www.datamintelligence.com/download-sample/pharmaceutical-microbiology-qc-testing-market
Pharmaceutical Microbiology QC testing encompasses key procedures designed to identify, quantify, and control microbial contamination in pharmaceutical and biopharmaceutical products throughout development and manufacturing. It is a cornerstone of quality assurance for sterile injectables, biologics, vaccines, and advanced therapies.
Key Developments
January 2026: Biotactical B.V. launched a 48-hour rapid sterility test using ATP bioluminescence, enabling pharmaceutical manufacturers to cut down the traditional 14–21 day sterility testing period and accelerate batch release.
January 2026: BIOMÉRIEUX completed the acquisition of Accellix, Inc., enhancing its pharmaceutical QC portfolio and strengthening rapid microbiology and advanced testing capabilities for improved contamination detection and regulatory compliance worldwide.
November 2025: BWT Holding GmbH unveiled the AQU Sense MB system for real-time microbiological monitoring in pharmaceutical water systems, providing results in approximately 20 minutes and facilitating faster contamination control during manufacturing.
Market Drivers and Opportunities
1. Regulatory Emphasis on Safety and Compliance
Stringent guidelines from agencies like the US FDA and other global regulatory authorities are catalyzing adoption of microbial QC testing to ensure sterility assurance, contamination control, and data integrity. Rapid and validated microbiological methods are increasingly encouraged under science‑based frameworks, creating significant growth opportunities.
2. Rise in Biologics and Advanced Therapies
The rapid expansion of biologics, vaccines, gene therapies, and sterile formulations has intensified the need for high‑throughput, accurate microbial testing solutions. This trend supports demand for both traditional and novel rapid QC platforms across manufacturing workflows.
3. Technological Innovation
Advanced technologies such as automated detection systems, next‑generation sequencing, IoT‑enabled monitoring, and real‑time microbial surveillance are driving operational efficiency and meet growing expectations for precision and speed in QC testing.
Market Segments
By test type, traditional/conventional testing dominated the global pharmaceutical microbiology QC testing market, holding the largest revenue share of 72% in 2025, while rapid testing is emerging as a key driver for future growth.
By Product: Instruments, Reagents & Kits, Accessories, Software, , By Test Type, Traditional / Conventional Testing, Rapid Testing
By Testing Workflow: In-Process Quality Control Testing, Finished Product Testing, Raw Material Testing, Utilities & Water Testing, Cleaning Validation Testing
By Technique: Growth-Based Techniques, DNA/RNA Amplification Techniques, Enzyme-Linked Detection Techniques, Cell Wall Component Detection, Metabolic Activity Detection, Immunological Assays, Others
By Detection Technology: Optical Detection, Fluorescence-Based Detection, Bioluminescence Detection, Impedance-Based Detection, Spectrometric Detection, Others
By Application: Bioburden Testing, Sterility Testing, Environmental Monitoring, Endotoxin Testing, Pyrogen Testing, Mycoplasma Testing, Microbial Identification, Others
By Sample Type: Finished Pharmaceutical Products, Active Pharmaceutical Ingredients (APIs), Excipients & Raw Materials, Packaging Materials, Process Water & Utilities, Air & Surface Samples
By End User: Pharmaceutical Manufacturers, Biotechnology Companies, Contract Manufacturing Organizations (CMOs), Contract Research Organizations (CROs), Academic & Research Institutes, Regulatory & Government Laboratories, Others
Request for Customized Sample Report as per Your Business Requirement:- https://www.datamintelligence.com/customize/pharmaceutical-microbiology-qc-testing-market
Market Regional Share
North America - Pharmaceutical Microbiology QC Testing
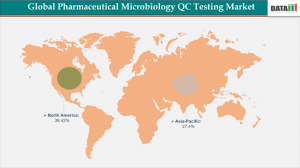
North America remains the dominant region in the global pharmaceutical microbiology QC testing market, securing the highest revenue share of 38.42% in 2025.
• Largest market due to a strong pharmaceutical and biopharmaceutical manufacturing base.
• High regulatory standards and FDA cGMP drive adoption of traditional and rapid microbial testing.
• Early adoption of automation and quick detection technologies accelerates batch release and contamination control.
United States - Market Outlook
• Leading producer of biologics, vaccines, and sterile drugs, fueling QC testing demand.
• Pharma and CDMOs increasingly use automated environmental monitoring, rapid sterility testing, and microbial identification.
• Integration of Industry 4.0 tools (automation, digital connectivity, analytics) can boost lab productivity by 50-100%.
Canada - Market Trends
• Growth driven by biologics, vaccines, and expanding contract manufacturing and testing services.
• Health Canada's adherence to GMP ensures continuous demand for sterility, bioburden, and environmental monitoring.
• Rapid microbiological methods are increasingly used in sterile production to improve efficiency and compliance.
Asia-Pacific - Fastest Growing Market
• Rapid pharmaceutical and biopharma expansion, stricter regulations, and rising exports drive growth.
• Investments in sterile drugs, vaccines, biosimilars, and cell/gene therapies increase QC testing demand.
• Adoption of automated and rapid microbial testing supported by compliance with FDA, EMA, and WHO GMP standards.
India – Market Outlook
• Emerging as a global pharma hub, producing over 60% of vaccines and 20% of generics.
• "Make in India" initiative boosts pharmaceutical and biopharma facility investments.
• Growth in sterile and biologic drug production drives demand for strict sterility, bioburden, endotoxin, and environmental monitoring.
China – Market Outlook
• Expansion of pharmaceutical and biopharma manufacturing fuels microbiology QC testing needs.
• Strengthened NMPA GMP enforcement and global compliance drive sterility, bioburden, endotoxin, and environmental monitoring.
• Domestic biopharma and CDMOs adopt automated QC testing; notable milestone: SGS Shanghai lab achieved FDA “Zero 483” review in Oct 2024.
Market Key Players
The competitive landscape of the Pharmaceutical Microbiology QC Testing Market remains concentrated with global leaders offering broad portfolios of instruments, reagents, rapid detection systems, and QC services.
Prominent key players include:
1. Thermo Fisher Scientific Inc.
2. BIOMÉRIEUX
3. Merck KGaA
4. BD (Becton, Dickinson and Company)
5. Danaher Corporation
6. Sartorius AG
7. Rapid Micro Biosystems, Inc.
8. Charles River Laboratories
9. HiMedia Laboratories
10. Hardy Diagnostics
Recent Developments - United States (2025‑2026)
February 2026: Charles River Laboratories expanded its microbiology QC testing services tailored for biopharmaceutical manufacturers, optimizing turnaround time for QC workflows.
January 2026: Thermo Fisher Scientific launched new automated microbial detection platforms designed to enhance both speed and accuracy within pharmaceutical QC laboratories.
Buy Now & Unlock 360° Market Intelligence:- https://www.datamintelligence.com/buy-now-page?report=pharmaceutical-microbiology-qc-testing-market
Recent Developments - Japan (2025‑2026)
February 2026: Sysmex Corporation introduced advanced microbial QC solutions for pharmaceutical production, emphasizing real‑time monitoring for contamination control.
January 2026: FUJIFILM Wako expanded its contract QC testing services in Japan to include rapid pathogen detection in pharmaceutical formulations, increasing testing capabilities and service breadth.
Conclusion
The Pharmaceutical Microbiology QC Testing Market is poised for accelerated growth driven by regulatory pressures, technological innovation, and an expanding pipeline of sterile and advanced biologic products. North America maintains market dominance, while Asia‑Pacific presents strong opportunities for future expansion. With continuous advancements in rapid microbial detection and quality control solutions, the market is expected to deliver substantial value to pharmaceutical manufacturers and contract testing organizations through 2033 and beyond.
Related Reports
Japan Microbiology Culture Market (2025)
MENA Microbiology Culture Market
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.